1
Introduction
In 2009, the Institute of Medicine (IOM) of the National Academies of Sciences, Engineering, and Medicine (the National Academies) published guidelines on weight gain during pregnancy. The report, Weight Gain During Pregnancy: Reexamining the Guidelines, concluded that there was insufficient evidence to support weight gain recommendations for women in higher classes of obesity1 or those who are underweight (body mass index [BMI] < 18.5 kg/m2) (IOM and NRC, 2009). However, the increasing prevalence of obesity among women of reproductive age means that more women with obesity are becoming pregnant and they are doing so at higher BMI values.2 As of 2022, nearly half of women of reproductive age (18–44 years) in the United States were classified as overweight, and 34 percent were classified as having obesity, with a growing proportion of women classified as having obesity Class III or above. These women are more likely than those classified with a healthy weight to gain weight outside the current guidelines for pregnancy (too little or too much). Both obesity and gaining excess weight during pregnancy may exacerbate the potential for adverse outcomes during the perinatal period.
To address the issues related to gestational weight gain (GWG), and to explore whether there is evidence to support updating the recommendations, the National Academies hosted a virtual workshop on September
___________________
1 The IOM and NRC (2009) report adopted the World Health Organization Class II (BMI = 35−39.9 kg/m2) and Class III (BMI ≥40 kg/m2) for higher classes of obesity.
2 The World Health Organization classifies BMI among adults as: underweight (< 18.5 kg/m2), normal weight (18.5–24.9 kg/m2), overweight (25.0–29.9 kg/m2), Class I obesity (30.00–34.99 kg/m2), Class II obesity (35.00–39.99 kg/m2), or Class III obesity (≥ 40.00 kg/m2).
4–5, 2025. This workshop was supported by the U.S. Centers for Disease Control and Prevention (CDC), the U.S. Department of Agriculture (USDA), and the Endocrine Society. The workshop focused primarily on women with obesity but also addressed the needs of underweight women. Speakers explored topics including emerging evidence on health outcomes related to prepregnancy weight and GWG, the role of dietary patterns in GWG and pregnancy-related outcomes, and the relationship of social and economic characteristics to prepregnancy BMI and GWG. This evidence, said Kathleen Rasmussen, chair of the planning committee, will inform the need to update the 2009 report. She noted that these guidelines are used in the United States by organizations such as the American College of Obstetrics and Gynecologists (ACOG) and the CDC.
Cindy Davis, National Program Leader for Human Nutrition at the USDA Agricultural Research Service (ARS), noted in her introductory remarks to the workshop that the mission of the USDA Human Nutrition Program is to define the role of food and its components in optimizing health throughout the life cycle for all Americans by conducting high national priority research. This workshop, she said, will explore how ARS scientists can address research gaps to advance understanding of maternal and child nutrition. Sacha Uelmen, Director of Professional and Clinical Affairs at the Endocrine Society, told participants that the Endocrine Society is committed to this critical aspect of women’s health and noted that the Society recently published a joint international guideline on pregnancy and diabetes.3
The workshop was planned by a committee according to a Statement of Task (Box 1-1). These proceedings summarize the presentations and discussions at the workshop. Chapter 1 sets the stage for the workshop, with background on the 2009 guidelines, the research response to the guidelines, a review of new evidence since 2009, and an example of similar guidelines from Brazil. Chapter 2 explores the challenges in pregnancy related to severe obesity and underweight, and Chapter 3 looks at the social and economic predictors of prepregnancy weight and GWG. Chapter 4 identifies potential approaches for mitigating the health effects associated with prepregnancy weight gain and GWG, including public health, data-driven, and clinical approaches. The workshop agenda is provided in Appendix A, and biographies of planning committee members and speakers in Appendix B. A scoping review that was undertaken to provide an updated review of the amount of available evidence on health outcomes related to weight gain during pregnancy among those in higher BMI categories as well as underweight, since the release of the 2009 guidelines is provided in Appendix C.
___________________
3 https://www.endocrine.org/clinical-practice-guidelines/preexisting-diabetes-in-pregnancy (accessed October 6, 2025).
BOX 1-1
Statement of Task
A planning committee of the National Academies of Sciences, Engineering, and Medicine will organize a 1-day public workshop that will explore the current state of evidence on perinatal health outcomes associated with weight gain during pregnancy. The workshop will feature invited presentations and discussions that may include, but are not limited to:
- The role of obesity prior to conception on perinatal outcomes,
- Maternal health outcomes associated with gestational weight gain by preconception BMI category,
- Fetal and infant outcomes during the perinatal period and later childhood,
- The role of total diet and dietary patterns in weight gain and pregnancy-related outcomes, and
- Prepregnancy BMI and perinatal weight gain as they relate to social and economic variables.
The planning committee will select and invite speakers and discussants and moderate the discussions. A workshop proceedings of the presentations and discussions at the workshop will be prepared by a designated rapporteur in accordance with institutional guidelines.
SETTING THE STAGE
In the 1970s, there was a single recommendation for GWG, said Monique Hedderson, Kaiser Permanente Northern California. The National Research Council report Maternal Nutrition and the Course of Pregnancy recommended that all women gain between 20 and 25 pounds during pregnancy (NRC, 1970). In 1990, the guidelines were updated and reflected the idea that GWG should vary depending on prepregnancy BMI (IOM, 1990). In 2009, the guidelines were updated again in three specific ways: there were recommendations for GWG based on prepregnancy BMI, recommended weight gain ranges by trimester, and an upper limit for GWG for those with obesity (IOM and NRC, 2009). While the changes to the 2009 guidelines were made in the context of a rising obesity epidemic, said Hedderson, the guidelines have remained unchanged for 16 years while the obesity epidemic has grown. When the first guidelines were published in 1970, the percentage of U.S. adult women with obesity was less than 20 percent (Figure 1-1). By the time the guidelines were updated in 1990, this had increased to 25 percent. By 2009, over 35 percent of women were obese, and the upward trend has continued since.
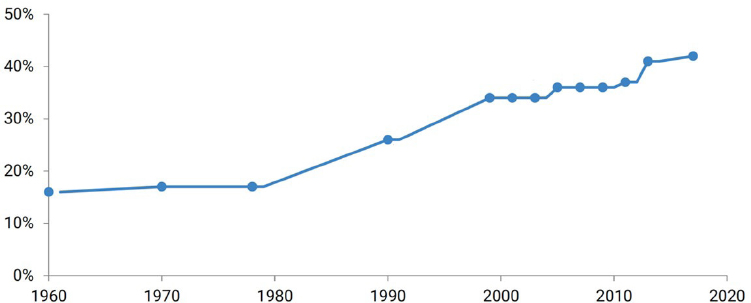
SOURCE: Adapted from presentation by Monique Hedderson on September 4, 2025; Fryar et al., 2016; Hales et al., 2020. Figure is courtesy of Lisa Bodnar.
Prepregnancy BMI
The percentage of pregnant women with a prepregnancy BMI classified as underweight or normal has decreased over time, while the percentage with a prepregnancy BMI of overweight or obese has increased (Figure 1-2). Hedderson noted that the largest relative increase has been among those with the most severe classes of obesity; there was a 14 percent increase in the percentage of women entering pregnancy with a BMI over 40. Unpublished data from Kaiser Permanente Northern California mirror these trends, said Hedderson, with increases in prepregnancy obesity across all obesity classes, and the largest relative increase among those with the most severe obesity. When these numbers are stratified by race, Hedderson said that “the disparities are striking.” The prevalence of prepregnancy overweight and obesity is most pronounced among Black and Hispanic women, although increases have been seen in all racial and ethnic groups (Wang et al., 2021). Hedderson noted that while Asian women have the lowest prevalence of overweight and obesity, the risks of adverse perinatal outcomes emerge at lower BMI thresholds in this population (Wang et al., 2021). In addition to racial disparities, Kaiser Permanente has also found disparities based on the Neighborhood Deprivation Index; women living in the most deprived neighborhoods have the highest rates of obesity while women in the least deprived neighborhoods have the lowest rates. Hedderson explained that obesity rates are influenced by neighborhood characteristics such as the built environment, access to healthy foods, and safe spaces for physical activity.
More women than ever—about one-third—are entering pregnancy with obesity. This matters, said Hedderson, because prepregnancy BMI is a risk
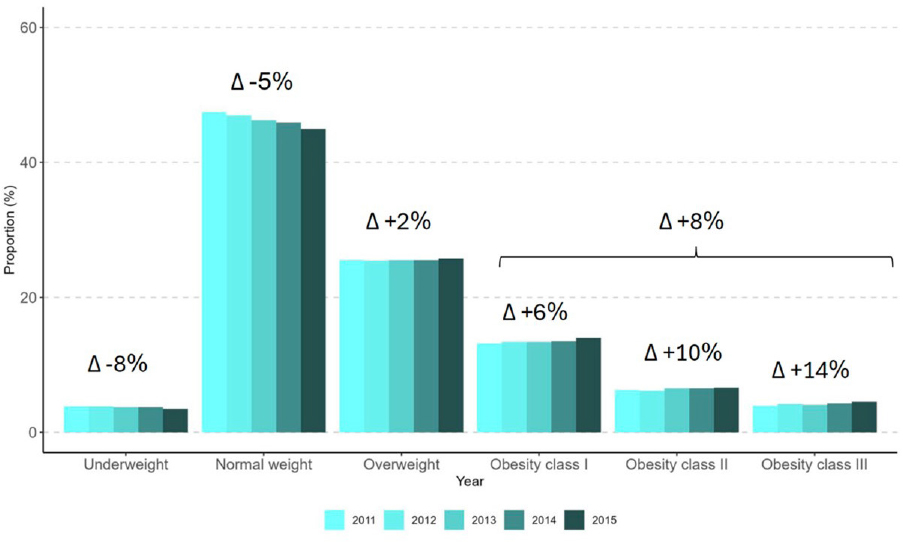
NOTE: BMI = body mass index.
SOURCE: Presented by Monique Hedderson on September 4, 2025; Deputy, 2018.
factor for maternal and infant adverse outcomes. A systematic review and meta-analysis of over 3 million pregnancies found a clear dose–response relationship between prepregnancy BMI and the risk of gestational diabetes, hypertensive disorders of pregnancy, cesarean birth, macrosomia, neonatal intensive care unit admissions, hypoglycemia, early postnatal infection, and other adverse outcomes (D’Souza et al., 2019). As would be expected from this relationship, said Hedderson, the rise in obesity rates has coincided with a rise in the incidence of adverse pregnancy outcomes. Studies have found increases in the rates of hypertensive disorders of pregnancy, low birth weight, and preterm birth (Freaney et al., 2022); severe maternal morbidity (Fink et al., 2023); and gestational diabetes (Shah et al., 2021).
Gestational Weight Gain
The 2009 IOM guidelines recommended that women gain different amounts of weight during pregnancy depending on their prepregnancy BMI (Table 1-1). A 2015 study examined how closely women were adhering to these guidelines (Deputy et al., 2015). It found that less than 50 percent of U.S. women were gaining weight within the IOM recommendations, and that adherence varied by prepregnancy BMI (Figure 1-3). Women who began pregnancy with BMIs in the underweight or normal weight categories were the most likely to gain weight within the recommended guidelines, although underweight women were also most likely to gain less than recommended. Women in the overweight and Class I obesity categories were the most likely to gain more than recommended, with over two-thirds of women exceeding the recommended GWG. Women with Class II and Class III obesity were less likely to gain above the
TABLE 1-1 2009 IOM Guidelines for Weight Gain During Pregnancy, by Prepregnancy BMI
| Prepregnancy BMI (kg/m2) | Total weight gain (lbs) | Mean rate of GWG (lbs/week)* |
|---|---|---|
| Underweight (< 18.5) | 28–40 | 1 (1–1.3) |
| Normal weight (18.5–24.9) | 25–35 | 1 (0.8–1.0) |
| Overweight (25.0–29.9) | 15–25 | 0.6 (0.5–0.7) |
| Obese (≥ 30.0) | 11–20 | 0.5 (0.4–0.6) |
NOTE: BMI = body mass index; GWG = gestational weight gain; IOM = Institute of Medicine; kg = kilograms; lbs = pounds; m = meters.
* 2nd and 3rd trimester rate. Recommendation for the 1st trimester is 1.1–4.4 lbs for all BMI groups.
SOURCE: Presented by Monique Hedderson on September 4, 2025; IOM and NRC, 2009.
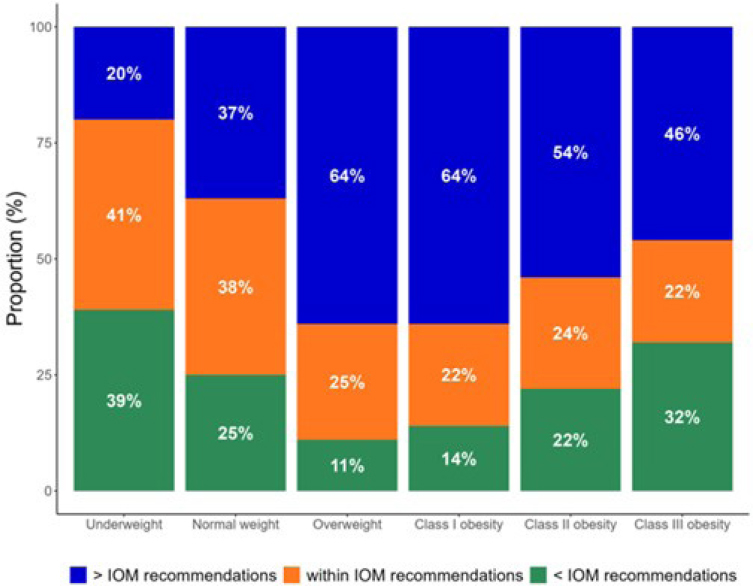
NOTE: BMI = body mass index; IOM = Institute of Medicine.
SOURCE: Presented by Monique Hedderson on September 4, 2025; Deputy et al., 2015.
recommendations, but more likely to gain less than the recommended amount. These data, said Hedderson, suggest that the guidelines are challenging for pregnant women to meet.
In closing, Hedderson summarized the information she had presented. The proportion of individuals entering pregnancy with overweight and obesity has increased over time, and there have been particularly steep increases in those with severe obesity. While these trends have occurred among all pregnant people, there are disparities by race, ethnicity, and socioeconomic disadvantage. Following the 2009 IOM guidelines for GWG, studies have demonstrated that most pregnant individuals are not meeting the recommended amounts for GWG, particularly those entering pregnancy with overweight and obesity.
REFLECTIONS ON THE 2009 GUIDELINES
Lisa Bodnar, University of Pittsburgh School of Public Health, served on the committee that produced the 2009 guidelines, and she shared her reflections on the process and what has happened since. The reexamination of the 1990 guidelines was prompted by several factors, she said: increases in rates and severity of obesity, changing demographics4 of the pregnant population, increases in chronic health problems before conception, excessive pregnancy weight gain in all BMI groups, and a growing recognition of the effect of excessive GWG. The committee used a conceptual framework to inform its review of the evidence and its recommendations; the framework reflected the different levels of influence on health and weight gain—individual, interpersonal, community and organizational, and policy and societal. The fact that there are numerous levels of influence, said Bodnar, means that interventions to affect weight must target multiple levels.
Bodnar shared several key findings that were influential in the development of the committee’s recommendations in the 2009 guidelines report (IOM and NRC, 2009). One finding was that prepregnancy BMI is more strongly related to adverse outcomes than GWG. Interventions to address this important risk factor can occur both before conception and in between pregnancies, said Bodnar. She explained that the committee’s decision to focus primarily on GWG was due to the fact that there are more opportunities for intervention during pregnancy than before pregnancy, since most women do not go to the doctor for preconception care. Another reason for focusing on GWG was a finding that many health outcomes are related to GWG. There was strong evidence that GWG had an effect on birth weight (both small and large for gestational age), preterm birth, child obesity, cesarean delivery, and postpartum weight retention. There was growing evidence of effects in other areas, but more data were needed; these areas included stillbirth, infant death, gestational diabetes, preeclampsia, breastfeeding, and longer-term health and obesity for both infant and mother. Another key finding, said Bodnar, was the need to account for the risks of both low and high GWG. Low GWG was associated with preterm birth and small-for-gestational age, whereas high GWG was associated with large-for-gestational age, cesarean delivery, and child obesity.
There were several areas in which more data were needed in order to support making recommendations, said Bodnar. First, there were inadequate data to stratify weight gain guidelines by severity of obesity. Second, there were too few weight gain intervention trials to provide information on whether and how weight gain can be modified. Third, there was a lack of data on important outcomes, such as the association between GWG and
___________________
4 As noted in presentation by Monique Hedderson on September 4, 2025.
preeclampsia and gestational diabetes, the effect of GWG on long-term health, and the link between GWG and rare but serious outcomes like maternal and infant mortality.
The committee wrestled with questions about which outcomes were more serious than others, how to reconcile the various risks and trade-offs, and what to do about areas with inadequate data, said Bodnar. What was needed, she said, was quantitative data that would allow a systematic balancing of risks of low and high weight gain for mothers and children—in other words, a formal decision analysis. This would have included utility values for all health outcomes, precise risk estimates for competing outcomes across the weight gain continuum, and quantitative methods to weight outcomes when recommendations have opposing effects. In the absence of this information, the committee relied on clinical judgment and qualitative assessments of data.
Despite the evidence gaps, said Bodnar, the guidelines were successful in many ways. They were adopted by ACOG and many international organizations. Materials were developed and disseminated for care providers, pregnant people, and people planning to become pregnant. Mechanisms to collect data on BMI and GWG were integrated into electronic health record systems and birth certificates, allowing for increased research and surveillance. The biggest accomplishment, said Bodnar, was the “giant research explosion” that occurred. The research community moved quickly to fill the knowledge gaps that were identified in the report; from 2009 to 2024, there were 10 times as many papers published on GWG than there were from 1990 to 2009.
REVIEW OF NEW EVIDENCE
The statement of task for this workshop (Box 1-1) called for an exploration of the current state of evidence on the associations between health outcomes, GWG, and preconception BMI, said Bodnar. To support this exploration, members of the workshop planning committee and National Academies staff conducted a scoping review of the literature.5 During deliberations for the 2009 report on GWG, said Bodnar, the committee concluded that there was insufficient evidence to stratify GWG guidelines by obesity class. The scoping review was intended to look for new evidence on health outcomes in relation to GWG, particularly evidence that supports stratification by class of obesity. Bodnar noted that the scoping review focused on the amount of evidence, not on summarizing the evidence itself. To be included in the scoping review, articles had to be peer reviewed and published in English between January 2008 and March 2025, and they had
___________________
5 The scoping review report is provided in Appendix C.
to include information on GWG in adult women, prepregnancy or early pregnancy BMI, stratification by BMI category, maternal outcomes, and infant outcomes. Bodnar noted that they excluded studies that were affected by reverse causality bias; that is, studies in which the exposure (total weight gain during pregnancy) could have been affected by the outcome (e.g., gestational diabetes). She observed that the vast majority of studies accounted for gestational age when making associations between GWG and outcomes; this is particularly important for outcomes associated with gestational age, such as preterm birth, stillbirth, and infant mortality.
Over 10,000 records were identified, and after screening for eligibility, 123 articles were included in the review. Of these articles, 97 included infant/child outcomes, 62 included maternal outcomes, and 36 included both maternal and child outcomes. Maternal outcomes included gestational diabetes, preeclampsia, gestational hypertension, cesarean delivery, induction of labor, operative vaginal delivery, postpartum weight retention, maternal morbidity, depression, and cardiometabolic disease. The majority of articles looked at maternal outcomes in the antepartum period (35) and intrapartum period (38), with fewer looking at outcomes in the first year postpartum (20) and more than 12 months postpartum (11). Infant/child outcomes included large-for-gestational age (LGA), small-for-gestational age (SGA), preterm birth, infant mortality, respiratory distress syndrome, neonatal hypoglycemia, obesity, allergies/atopy, and intellectual development disorders. Similar to the articles looking at maternal outcomes, most articles looked at shorter-term outcomes; the majority examined outcomes during the fetal/birth period (73), with fewer examining the first year of life (25), 1–3 years (15), 4–8 years (19), and 9–18 years (9). A number of articles looked at outcomes by class of obesity, said Bodnar. These articles looked primarily at short-term outcomes such as cesarean delivery, LGA and SGA, and preterm birth. While less common, she said, there are some articles on longer-term outcomes including infant death, child obesity, and postpartum weight retention.
In summary, said Bodnar, the scoping review revealed that there is a large body of data on GWG and short-term outcomes. While the number of studies on long-term outcomes has increased, more work is needed. Research on outcomes by class of obesity has been limited but is growing. There is a particular need, she said, for research that uses the same cohort to look at short- and long-term outcomes for both mother and child.
RESPONSE TO THE 2009 GUIDELINES
As Bodnar described, there has been growth in the evidence base on GWG and health outcomes since the 2009 guidelines were developed, said Leanne Redman, Pennington Biomedical Research Center. Redman
presented information on some of the new evidence; in particular, she focused on key advances in intervention-based studies, observational studies, and personalized medicine.
Lifestyle Interventions in Expectant Moms (LIFE-Moms)
Pregnancy is a critical life stage that can positively or negatively affect the future risk of cardiometabolic disease for both the pregnant individual and their offspring. Interventions during pregnancy may be one way to break this intergenerational transmission of disease, said Redman. Soon after the 2009 guidelines were released, the National Institutes of Health (NIH) published a funding opportunity calling for research on lifestyle interventions during pregnancy for individuals living with overweight or obesity. Lifestyle Interventions in Expectant Moms (LIFE-Moms) was born out of this funding: a consortium of seven clinical sites and a coordinating center conducted a study of lifestyle interventions for 1,150 pregnant women with overweight and obesity (Clifton et al., 2016). Each clinical site focused on a different type of lifestyle intervention, said Redman, but all sites used common inclusion–exclusion criteria and collected uniform data on key outcomes to enable data aggregation and comparison. The interventions being tested included nutrition interventions, meal replacements, telehealth, and home visitation.
The primary outcome measured was GWG, both per week and total. Compared to the standard of care, women in the intervention groups had reduced GWG per week, and a significant reduction in total GWG of about 1.6 kilograms. Collectively, said Redman, the interventions improved adherence to the 2009 guidelines, but a large proportion of participants still experienced excessive weight gain. Sites collected other data on both maternal and fetal/infant outcomes, including gestational diabetes, preeclampsia, cesarean delivery, and preterm birth. While there were some differences between the intervention group and the standard of care group, none of the differences was statistically significant. Redman pointed out a few caveats that may have limited the study’s findings. First, all interventions were required to enroll participants prior to the 16th week of pregnancy; the trials, therefore, could not answer questions about early intervention versus later intervention. Second, the trials were not pragmatic because individuals with preexisting conditions such as hypertension or diabetes were excluded. Finally, the interventions were not personalized at the individual level. That is, participants in a specific intervention received global recommendations for physical activity or nutrition, rather than recommendations based on their own specific circumstances.
For other evidence on lifestyle interventions, Redman told participants about a systematic review published in 2021 for the U.S. Preventive
Services Task Force (USPSTF). The review included 68 studies that looked at counseling and behavioral interventions for healthy weight and weight gain during pregnancy; collectively, the studies had almost 26,000 total participants. These interventions, said Redman, were found to result in an absolute risk difference of -7.6 percent for GWG, or around 1 kilogram. In addition, the absolute risk difference was -1.6 percent for gestational diabetes, -2.4 percent for emergency cesarean, -1.9 percent for macrosomia, and -1.3 percent for LGA. Based on these data, USPSTF made a new recommendation that all pregnant individuals be offered effective behavioral counsel-ling interventions to promote healthy weight gain and to prevent excess GWG. Redman noted that the recommendations acknowledged that it was unknown what the ideal duration or intensity of interventions should be.
Observational Studies
Since 2009, there have been advances in evidence from observational studies about how to advise women on dietary changes during pregnancy. The 2009 committee relied on the Dietary Reference Intake (DRI) report on macronutrients from 2005 (IOM, 2005), which stated that individuals need no extra calories in the 1st trimester, 340 extra calories per day in the 2nd trimester, and 450 extra calories per day in the 3rd trimester. However, the DRI report noted that these amounts are for women who are normal weight before pregnancy; women with overweight or obesity may need fewer calories. Redman and her colleagues conducted an observational study in pregnant women with obesity (Most et al., 2019). After the women gave birth, the researchers classified their total weight gain, according to the 2009 recommendations, as recommended, excess, or inadequate (Figure 1-4). Individuals who gained the recommended amount of weight did so by being in a negative energy deficit; compared to prepregnancy caloric intake, they consumed around 120 calories less per day throughout the 2nd and 3rd trimester. Women who gained an excess amount of weight during pregnancy were consuming around 180 extra calories per day, which is a relatively small amount. Redman pointed out that if women with obesity followed the 2005 DRI recommendations, they would be taking in 340 to 450 extra calories a day; to gain weight within the recommended range, they would need to take in either the same or fewer calories than before pregnancy. This demonstrates, said Redman, that pregnant individuals need personalized dietary guidance rather than blanket advice.
Another trial randomized 118 pregnant women with obesity and prescribed a 30 percent calorie reduction for the intervention group (Vesco et al., 2014). Women in the intervention group had a total weight gain of 5 kilograms, compared to 8.4 kilograms in the control group. Excess weight
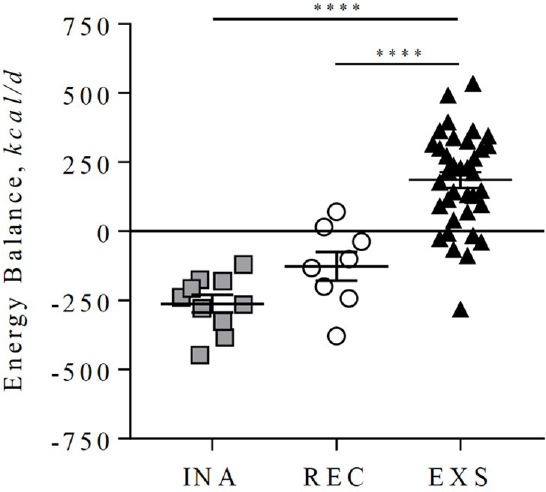
NOTE: EXS = excess; INA = inadequate; REC = recommended.
SOURCE: Presented by Leanne Redman on September 4, 2025; Most et al., 2019. CC BY 4.0.
gain was reduced by 50 percent, which Redman said was “remarkable.” Perinatal outcomes were also improved with the dietary restriction.
Personalized Medicine
The DRI report was updated in 2023, said Redman, with new recommendations that gave estimated energy requirements for pregnant people; the recommendations take into account activity level, stage of pregnancy, age, height, and weight. These guidelines are helpful for providing personalized dietary intakes for pregnant women, she said, and there are other ways to personalize the care of pregnant women with obesity, based on their individual circumstances. Obesity is not a single factor that affects perinatal health but is part of a constellation of interrelated factors. Flanagan and colleagues (2021) found that some individuals have metabolically healthy obesity (MHO), in which they have high BMIs but no other risk factors for metabolic syndrome, while others have metabolically unhealthy obesity (MUO), in which they have high BMIs plus two or more risk factors such as high blood pressure or hyerperlipidemia.
During pregnancy, individuals with MUO actually gain less weight than individuals with MHO, but they have larger increases in triglycerides, a higher prevalence of gestational diabetes, and their infants are born heavier and with more body fat. Based on these findings, Redman said that there is room to improve the personalization of lifestyle interventions for pregnant women, and specifically, to make more precise dietary recommendations for women with high BMI. There is a need, however, for pragmatic trials that look at the implementation of interventions in the community. She noted that the LIFE-Moms study showed that intensive interventions can reduce excess GWG, but that the reduction of 1.5 kilograms was still not enough to change perinatal outcomes. Moving forward, said Redman, all pregnant women should be offered weight management support, but there is a need to develop and test more precise approaches that target individuals rather than groups.
BRAZILIAN GESTATIONAL WEIGHT GAIN GUIDELINES
Since the 1980s, Brazil has been using tools from other countries to monitor GWG, said Gilberto Kac of the Federal University of Rio de Janeiro. These tools were neither validated nor properly tested for use in Brazil. Around 2005, Kac approached the Ministry of Health and requested that it convene a working group to develop guidelines specifically for Brazil. The initial results were published in 2009 (Kac et al., 2009); GWG guidelines were finally published in 2022. Kac told participants about the process and thinking behind these guidelines.
The Brazilian guidelines include GWG charts, which Kac explained are easy for health providers to explain and for pregnant individuals to understand (Table 1-2). These charts are commonly used in other fields, serve as a useful screening tool, and allow for continuous monitoring of GWG. To develop the charts, researchers led by Kac harmonized data from the Brazilian Maternal and Child Nutrition Consortium (BMCNC), using 21 studies representing more than 7,000 pregnant individuals (Carrilho et al., 2020). They used a prescriptive approach, said Kac, including only individuals without preexisting conditions, without adverse outcomes during pregnancy, and who gave birth to healthy children. Centile charts were developed for four prepregnancy BMI categories, from week 10 to week 40 of pregnancy (Kac et al., 2021). Next, the researchers analyzed the data to find the range of GWG that was associated with the lowest risk of maternal and infant outcomes. There were no good quality studies that looked at maternal and neonatal outcomes in the same dataset, he said, so they used data on postpartum weight retention from the Brazilian Food and Nutrition Surveillance System along with data on SGA, LGA, and preterm birth from the BMCNC and Birth in Brazil study (Carrilho et al., 2023).
TABLE 1-2 Brazilian Gestational Weight Gain Guidelines Compared to IOM 2009 Guidelines
| BMI category | Percentiles | Brazilian GWG ranges at 40 weeks (kg) | IOM 2009 (kg) |
|---|---|---|---|
| Underweight | P18 - P34 | 9.7 - 12.2 | 12.5 - 18.0 |
| Normal weight | P10 - P34 | 8.0 - 12.0 | 11.5 - 16.0 |
| Overweight | P18 - P27 | 7.0 - 9.0 | 7.0 - 11.5 |
| Obesity | P27 - P38 | 5.0 - 7.2 | 5.0 - 9.0 |
NOTE: BMI = body mass index; GWG = gestational weight gain; kg = kilogram.
SOURCE: Presented by Gilberto Kac on September 4, 2025.
Kac and his colleagues used a statistical method proposed by Hutcheon and Bodnar (2014) to analyze the data and identify GWG ranges where the risk of adverse outcomes was minimized. Kac noted that the direction of outcomes for mother and infant is not always aligned; for example, the GWG that was associated with the lowest risk of neonatal outcomes corresponded to a higher probability of excess postpartum weight retention. Experts from a variety of disciplines convened to discuss the proposed GWG ranges, weigh the risks, and achieve consensus on ranges for GWG. The 2022 guidelines have some overlap with the IOM 2009 guidelines but are more restrictive, particularly the GWG recommended for underweight women and the upper limits for normal weight, overweight and obesity.
Since the publication of the guidelines, said Kac, the Brazilian Obstetrics and Gynecology Federation has released a position statement in support (Surita et al., 2023), and tools have been developed to help providers and patients follow the guidelines. The development of these guidelines was an unprecedented initiative in a middle-income country, said Kac. The process was methodologically rigorous and resulted in valuable tools for monitoring maternal nutritional status during pregnancy and preventing adverse outcomes. Kac observed that using the IOM 2009 guidelines or others from high-income countries would have been inappropriate for Brazil and could have led to an increased risk of adverse outcomes such as excessive postpartum weight retention. Kac said that he is currently working with the World Health Organization to develop global GWG charts and to identify thresholds on the charts that define optimal weight gain ranges.
WEIGHTING THE SERIOUSNESS OF OUTCOMES
As other presenters have discussed, both high and low GWG can increase risks of poor pregnancy outcomes, said Jennifer Hutcheon, University of
British Columbia. Outcomes associated with low GWG include stillbirth and SGA, and outcomes associated with high GWG include LGA, child obesity, cesarean birth, and preeclampsia. Some of these outcomes are more serious than others, said Hutcheon; for example, stillbirth is a far more serious outcome than cesarean delivery. She argued that guidelines should take these differences in seriousness into account when identifying the range of pregnancy weight gain that minimizes risks of adverse outcomes. To do so, the committee that developed the 2009 guidelines commissioned a quantitative analysis of risk trade-offs. The analysis obtained estimates of the risks of infant mortality, excess maternal postpartum weight retention, and childhood obesity, by category of GWG. The authors applied a health utility value to each outcome (or a “seriousness weight”), and then identified weight gain values that maximize quality-adjusted life years.
Unfortunately, said Hutcheon, health utility values were not available for many of the important maternal and child health outcomes, and estimating the effect of these outcomes on health was too speculative given available data at the time. The committee noted this challenge in its report, and called on NIH and other agencies to fund research that would (1) assess the utility value associated with short- and long-term health outcomes that are associated with GWG, and (2) use these values in epidemiologic studies that are trying to link GWG with health outcomes to identify the range of GWG that best balances the risks of high and low gain.
Hutcheon worked with Bodnar and other colleagues to respond to this call for research. Using purposive sampling to obtain a group that reflected the diversity of the United States, they conducted an online Delphi panel of 82 pregnant or postpartum individuals and 84 clinical or public health perinatal health professionals (Bodnar et al., 2021). This panel was given a list of 11 health outcomes that are linked with GWG and was asked to rate the seriousness of each from 0 (not serious) to 100 (most serious). After a three-round process that involved discussion and feedback, the participants decided that the most serious outcomes were infant death, stillbirth, preterm birth, and preeclampsia, while the least serious were SGA, LGA, and cesarean birth. Although the process was not aimed at achieving consensus, there was a high degree of agreement among care providers and patients, said Hutcheon.
The next step was to apply these values to analyses seeking to establish GWG ranges associated with the lowest risks of maternal and child health outcomes. Hutcheon and her colleagues conducted a series of studies, both for individuals with normal weight and with overweight or obesity (Bodnar et al., 2024a,b). The aim of the studies was to identify the range of pregnancy weight gain that minimizes the risks of a composite outcome of 10 adverse maternal and child health outcomes, weighted for their seriousness, and to do so by prepregnancy BMI category. Using data from a
heart health study that included 2,344 nulliparous pregnant individuals, the researchers identified pregnancies that experienced any of 10 adverse outcomes and assigned a “seriousness score” to the pregnancies based on the values established in the Delphi process. The researchers conducted statistical analyses that modeled the risks of this weighted composite outcome along the GWG continuum. Hutcheon noted that it was important to look at the continuum of GWG, rather than using predefined categories, because it allows for estimation of risks along each weight gain increment and identification of optimal cutoff points.
There were two key findings in this research, said Hutcheon. First, weighting individual outcomes for their relative seriousness matters. When the seriousness of outcomes is taken into account, the risks of higher weight gain become more pronounced. She explained that common outcomes with higher weight gain, such as preeclampsia and obesity, were rated more serious than common outcomes associated with lower weight gain, such as SGA. The second key finding was that weight gain below current recommendations was not linked with increased risks. The upper limit of the 2009 recommendations aligns quite closely with the inflection point at which risks increase, she said, but there is not a meaningful increase in risk below the lower limit of the recommendations. This pattern was also seen in individuals with overweight and obesity.
Further, said Hutcheon, when the analysis was applied to a large population-based cohort of over 15,000 pregnant women with obesity, the conclusions were similar (Johansson et al., 2024). Weight gain above the recommendations was associated with greater risk for women in all obesity classes, and weight gain below the recommendations had no increased risk. In fact, for women with Class III obesity, weight gain below current recommendations was protective against adverse outcomes. These findings, she said, support the reevaluation of the lower limit in the 2009 guidelines. The current limit may be too high, and it may be appropriate to remove the lower limit altogether for women with obesity.
ROLE OF DIET IN WEIGHT GAIN AND PREGNANCY-RELATED OUTCOMES
Dietary patterns can be defined as “the quantities, proportions, variety, or combinations of different foods, drinks, and nutrients in diets, and the frequency with which they are habitually consumed,” said Christine Garner, Texas Tech University Health Sciences Center. Examining dietary patterns is a way of capturing the complexity of diet and interrelationships between foods; further, dietary patterns are more consistent over time than intake of individual nutrients. One way that dietary patterns in the United States are measured is the Healthy Eating Index (HEI). Garner explained that this tool
measures alignment between a person’s intake and the Dietary Guidelines for Americans (DGA) for a variety of components, including fruits and vegetables, whole grains, dairy, and added sugars. The highest score possible is 100, representing the highest-quality diet; the average score for Americans in the most recent DGA report was 59. There are other score or index-based tools for assessing dietary patterns, said Garner, as well as data-driven methods. Index-based tools allow for comparison between studies, she said, whereas data-driven dietary patterns can be harder to compare.
Pregnancy outcomes that have been studied in relation to dietary patterns include gestational diabetes, hypertensive disorders of pregnancy, gestational age at birth, and birth weight. A set of systematic reviews published in 2019 and 2024 (Abrams et al., 2024a,b; Andres et al., 2024; Byrd-Bredbenner et al., 2024; Raghavan et al., 2019a,b) analyzed these studies to determine associations between dietary patterns and perinatal health outcomes. The analyses found that diets that were higher in vegetables, fruit, whole grains, nuts, legumes, fish/seafood, and vegetable oils, and lower in red/processed meats, fried foods, added sugars, and saturated fats had a protective effect on outcomes. The 2019 reviews looked at both preconception dietary patterns as well as dietary patterns during pregnancy and found that preconception diet had a protective effect for gestational diabetes; evidence was more limited on the effect of preconception diet on hypertensive disorders. Protective dietary patterns during pregnancy were associated with reduced risk of preterm birth, but there was insufficient evidence about the effect on birth weight. The 2024 reviews served as an update to the 2019 reviews, said Garner, but they did not look at preconception dietary patterns. These reviews found that protective dietary patterns during pregnancy were associated with reduced risk of gestational diabetes and lower risk of SGA; evidence was insufficient for hypertensive disorders. Interestingly, said Garner, the updated reviews found that dietary patterns during pregnancy were not associated with the risk of preterm birth, in contrast to the conclusions drawn in 2019.
Garner noted that there are a number of limitations in the current evidence on the association between dietary patterns and perinatal outcomes. First, much of the research has been conducted in other countries, with populations that are not representative of the U.S. population in terms of race, ethnicity, income, access to health care, and other socioeconomic variables. Second, many of the studies have been insufficiently powered for the outcomes of interest, and there was considerable heterogeneity in controlled variables such as BMI, energy intake, gestational weight gain, and family history. Third, some studies reported outcomes in nonstandardized ways or used self-report of outcomes; in particular, birth weight outcomes were inadequately standardized for gestational age and sex. Finally, said Garner, there are limitations in how dietary patterns are assessed. Many
studies measured diet at one point in time, which could have been months or years prior to conception, others measured diet at the same time as they measured outcomes, and some did not specify the timing of dietary measurement. Garner said that there is a need for stronger, well-designed studies with larger, more diverse samples.
Dietary patterns need to be measured at multiple time points before and during pregnancy, using comparable measurements of diet (e.g., HEI). Based on the available evidence, she said, diet quality does appear to have an effect on perinatal outcomes, but further study is needed.
This page intentionally left blank.



















