Understanding and Preventing Violence, Volume 2: Biobehavioral Influences (1994)
Chapter: STUDIES INVOLVING HORMONES AND "AGGRESSION"
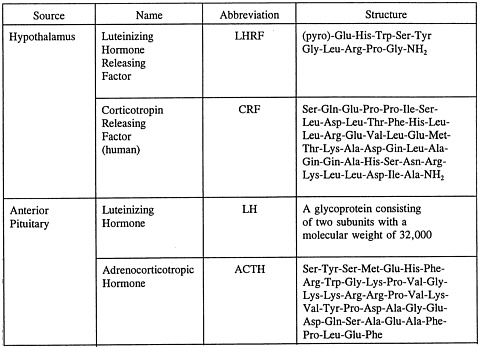
(hypothalamus-pituitary-adrenocortical axis, which includes corticopin releasing factor (CRF), ACTH, and adrenal steroids) axes. One should recognize, however, that other hormones (e.g., those of the thyroid gland and adrenal medulla; see Valenti and Mainardi, 1989, and Hucklebridge et al., 1981) can also be implicated in changes in violence and aggression (see Figure 3 for basic biochemistry).
A brief note on the respective significance of the different levels in the brain-pituitary-gonadal axis as they relate to violent and aggressive behavior might be appropriate here. Associations between hypothalamic LHRF and violence could be interpreted as the most neural component of the HPA axis directly influencing the CNS, or they could be regarded as suggesting that the axis is activated or deactivated in such responses. If LH is correlated with violence or aggression, it could suggest that this pituitary gonadotropin controls behavior before the major effects of the HPA system on gonadal function are established (this has been the interpretation favored in some seasonally breeding animals), or it might indicate that the feedback mechanisms of steroids are accentuated or suppressed. Correlations between androgens or estrogens and behavior are also capable of being interpreted in several ways (e.g., "direct" effects of hormones on violence via neural actions of steroids, or reflections of changes in the hypothalamus and/or pituitary).
STUDIES INVOLVING HORMONES AND "AGGRESSION"
The literature relating hormones to aggressive behavior has been reviewed on several occasions (see Brain, 1977-1981). One striking feature of this enormous body of data is that the sheer diversity of approaches and methodologies used makes extracting generalities from the data exceedingly difficult. These earlier reviews confirm that the topic of hormones and aggression in vertebrates can be effectively subdivided into
-
studies on the effects of early hormonal "programming" of adult aggressiveness;
-
direct effects (presumably via the CNS) of endocrine manipulations on fighting and threat;
-
indirect effects (presumably via changed social signals, etc.) of endocrine manipulations on fighting and threat;
-
hormone-aggression correlations; and
-
influences of fighting on endocrine function.
The types of manipulation involved in items (2) and (3) can be referred to as exogenous modifications. These are produced by
-
surgical removal of the endocrine glands;
-
injection of hormone in a solution at a variety of sites (e.g., subcutaneous, intramuscular, intracerebral);
-
implantation of hormone as a solid phase (as crystalline hormone or with a material such as Silastic) at a variety of sites (see above);
-
use of antihormones or blocking agents; or
-
transplantation of hormone-secreting materials (these may be functional endocrine glands or hormone-secreting tumors).
Sometimes treatments are used in combination, as in classical replacement therapy (surgical removal of the gland followed by injection of the hormone associated with the structure). One should note that all manipulations alter more than one component of the endocrine system. Hormones do not function in vacuo but operate as part of a complex integrated physiological system.
Endogenous changes are likely implicated in type (4) studies. Here one may use the following indicators of endocrine activity
-
organ weight,
-
histology,
-
morphometry,
-
histochemistry,
-
bioassay,
-
fluorometric assay,
-
competitive protein binding assay,
-
radioimmunoassay, or
-
enzyme immunoassay.
Measurements may be applied to tissues, plasma, or even saliva. Assays are generally judged on the basis of three criteria, namely,
-
sensitivity (the amount that can be detected);
-
reproducibility (the ability to get the same result twice running); and
-
specificity (the ability to measure only the particular hormone).
On these criteria, radioimmunoassay, enzyme immunoassay, and some of the new flourometric assays may be judged the most accurate current means of measuring hormones, but it must be stressed that all methodologies seem to have utility under particular
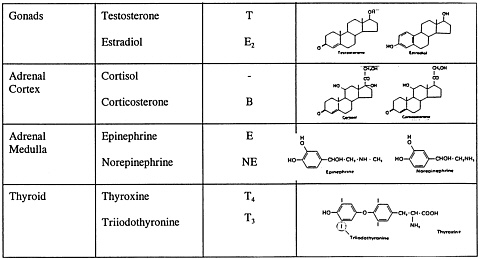
FIGURE 3 Structures of some hormones that appear to be important in aggression research. NOTE: The structures of LHRF, CRF, and ACTH are the sequences of amino acids; ACTH is actually 39 residues long, but only 1–24 are essential for the actions on the adrenal cortex (sequence 4–10 is shared by MSH and may have extra-adrenal actions on the brain); T3 has one iodine missing at the position indicated.
circumstances. For example, organ weights or histology may prove useful when the stress of obtaining blood samples is likely to confound the measurements or when the amount of material is very small. It is obvious that the concentrations of hormones at receptors are more likely to be of relevance to ongoing behaviors (including aggression) than changes in serum or plasma concentrations. This having been said, there are a variety of technical and ethical problems that make the obtaining of such data in clinical studies inherently improbable (at present). The best that can currently be achieved in human studies is to examine a range of values and to look at the time courses of these variations in detail.
One must note that a wide variation of sophistication is evident in the work on hormone-aggression correlations emanating from different specialties. A personal view is that modern plasma and saliva (these are especially useful because they reflect unbound hormone and the samples are obtained noninvasively with little associated stress) assays of multiple circulating hormones in humans are most appropriate, whereas attempts to estimate receptor dynamics in the brains of animals are of the greatest utility (these enable one to see how the hormones act at the level of the receptor).
It now seems unlikely that one will be able to show clear relationships between aggression and a single (even if immensely accurate) determination of the plasma value of a single hormone. One may argue that it is important to know
-
whether the titer is increasing or decreasing and whether the factor is elevated or depressed with relation to a fluctuating baseline;
-
the distribution of the hormone in the different body compartments (e.g., plasma and target tissues);
-
the extent to which the receptor (hormone-receiving points on the cell's membrane or within the cell) is occupied by bound hormone and the amount of such receptor (receptor populations can be altered by hormones); and
-
interactions with other hormones (many hormones change other components of the endocrine system); and
-
whether hormone production shows estrous, seasonal, or circadian rhythms.
Brain (1989c) has recently (in advocating an ethoexperimental approach to the study of relationships between hormones and behavior) detailed some of the complications that have become apparent
to workers in this area. Initially, hormones were assumed to directly influence the expression of a specific behavior (e.g., aggression). In this schema, the endocrine gland is viewed as functioning in isolation, and measures of its output are correlated with a specific behavioral measure. There has, in such cases, been a tendency to assume that the hormone produces the behavior by acting on the CNS.
As the vascular system carries hormones throughout the body, they may be picked up and change processes in a variety of structures that modify behavior. Although motivational changes (mediated via the CNS) are recognized as being one means of expressing hormonal action, these chemicals may modify the production of social cues and/or the sensory systems that detect such factors. Such effects may be expressed in a variety of sensory systems including the somatosensory, visual, olfactory, and auditory modes. There is good evidence that hormones can change the responsiveness of female mammals to male odors and that such sensory input has a powerful impact on social behaviors. One must also stress that other endocrine glands can be targets for the actions of hormones. The situation may be complicated by emphasizing this ''cross-talk" between endocrine glands (e.g., the effects of the gonadal system in mammals on the adrenal cortex and vice versa) and by recognizing that the altered target tissues may express their actions on behavior in rather different ways. It is rare for a single modality or mechanism to be employed. Rather than specifying a need to examine phenomena such as "cross-talk" in terms of their impact on aggression, it is simply advocated that one remember that a complex interactive system is involved. The actuality strongly suggests that one should look for multiple endocrine correlates of behavior, recognizing that some will be secondary consequences of other changes.
Another complication is the fact that metabolic conversions may transform initially secreted or applied hormones (in a variety of locations, including the blood and neural sites) to a range of compounds. Testosterone, for example, can be aromatized (chemically converted) to 17 ß-estradiol in certain neural locations and reduced to 5a-dihydrotestosterone in other neural locations and peripheral androgen-dependent tissues (e.g., the seminal vesicles; see Figure 4). Adrenal steroids (because of their chemical similarities) are also often interconverted.
A further complication is that one rarely sees single behavioral responses in isolation. Behavioral elements (which may be individual actions or broad categories of behavior) not only can
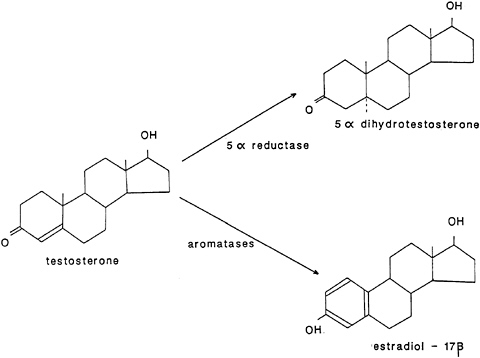
FIGURE 4 Conversion by enzyme systems in different targets of testosterone to 5a-dihydrotestosterone and estradiol-17ß.
interact (e.g., if an animal is showing fearful behavior, it is unlikely to explore) but can be influenced by a variety of hormone-target relationships, some of which can be common to several behaviors, and others exclusive.
The final complication introduced into the scheme is shown in Figure 5, reflecting the obvious fact that there are feedback relationships between many of the subprocesses linking endocrine gland activities to behavioral elements. It is well documented that behavioral experiences can have profound physiological repercussions (see Brain and Benton, 1983). Further, it is difficult to generalize across species and test situations or to infer common underlying mechanisms (Brain, 1979a; Miczek and Krsiak, 1981). Brain et al. (1983) have shown that the radically different laboratory tests used to assess murine aggression produce very different pictures when common manipulations of the gonadal system are attempted. It seems likely that these tests tap different mixtures of motivations (see earlier). Brain (1981) has emphasized the complex interplay between endocrine glands and the variety of target tissues that must be considered in any investigation
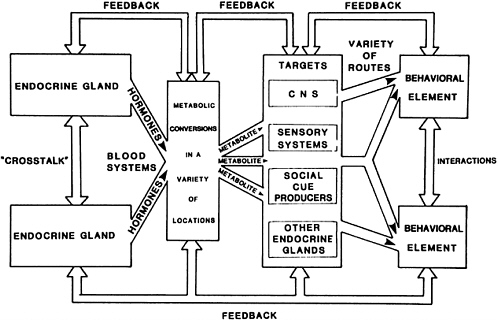
FIGURE 5 Associations between hormones and behavior stressing "feedback" relationships at many points in the scheme. Experiences obtained via behavioral expression can modify targets of hormonal action (especially the CNS) change metabolic conversion rates, and alter secretion of endocrine glands. SOURCE: Brain (1989c). Reprinted by permission of Kluwer Academic Publishers.
of hormonal involvement in aggression. It is certainly hard to assess the roles of hormones in aggression because this area of research involves a great number of "strong" and "weak" correlations between a difficult behavioral concept (aggression) and the complex and subtly integrated endocrine system (Brain, 1977).
One of the current difficulties of implicating hormones in agonistic behavior is that much of the accumulated literature in behavioral endocrinology is based on the traditional approaches of physiological psychology (see Carlson, 1977). Here, relatively crude (generally pharmacologic) manipulations of the endocrine system are usually attempted via surgery, implantation, or injection, and the consequences of the modification related to aggression are determined in restricted situations. For example, large doses of testosterone have been given to male chicks soon after hatching, and the effects on their aggressive responding to a hand have been assessed (Andrew, 1975). Sigg et al. (1966) were unable to demonstrate any action of repeated injections of 2 International Units per kilogram of thyroid stimulating hormone (TSH) on social conflict in male hypophysectomized (with the pituitary gland removed) mice. Suchowsky et al. (1969) claimed that estradiol, when given repeatedly at high doses to pairs of male animals, inhibited social conflict in intact and castrated albino mice subjected to a series of tests. The estrogen clearly modifies odor characteristics in such subjects. There are, in direct contrast, fertile developments in ethopharmacology, where a tradition of inclusive measurement of behavior is undertaken in animals who generally have options that are less restricted than those seen in traditional laboratory tests for aggression. Further, a current trend is to look for alternative explanations of hormone actions on behavior to those interpreted as "simple" changes in aggressiveness. A more ethoexperimental approach to behavioral description should enable us to assess the important or fundamental behavioral correlates of hormonal changes.
It would also make sense to use the recently developed, less invasive, sensitive assay techniques to measure as many different aspects of hormonal function as are feasible (including changes in binding site populations as well as titers of hormones). A second gain would follow from assessing behavior in more sophisticated situations. It is, naturally, impossible to show the behavioral impact of a particular hormonal manipulation in an inappropriate environment—many of the effects of hormones important to animals in the wild may only be apparent in more complex, familiar environments where the animal has the possibility of expressing
behavior in context. Progress may also depend on a willingness to be more concerned about the temporal relationships between endocrine changes and behavior. Much of what has already been described in the physiological psychology relates to the acute correlation of hormonal change with behavior, whereas it might be more fruitful to look at the organism more chronically and attempt to relate patterns of endocrine fluctuation to life events (e.g., onset of mating or assumption of a territorial habit). It should be possible to blend a reasonable degree of scientific rigor with richer and more flexible arenas for studying behavioral expression.
When commenting on the available literature, it seems worthwhile to stress an earlier claim (Brain, 1981) that it would not be especially surprising if all hormones alter some aspect of aggression in a particular species or a specific situation. As mentioned earlier, hormones can have very diverse actions and can change behavior in a variety of ways. Consequently, any modification of the endocrine environment can have consequences (e.g., altered brain architecture, increased body size, altered odor characteristics, or varied availability of energy substrates) that can change the probability of recording activities capable of being judged violent or aggressive. This having been said, certain hormone-behavior correlations are stronger than others. There is little doubt that the hormones associated with the HPA and HPG axes are more likely to have lasting impacts on behavioral development and concurrent behavioral expression than most other endocrine factors. At this state of our knowledge, perhaps only the hypothalamic-pituitary-thyroid axis, adrenomedullary hormones, and hormones that produce large changes in blood sugar levels (e.g., insulin and glucagon) are worthy of additional consideration. It is my belief that new studies in this area will tap a wider range of endicrine measurements and that they will be especially directed to the so-called metabolites of peptide and steroid hormones. One will also have to pay considerably more attention to changes in binding site populations in receptors, and to the rates of metabolic conversion and the clearance rates of hormones and their products.
The basic subdivisions of material referred to earlier are used in the following sections, with material on nonprimate vertebrates, infrahuman primates, and humans being presented separately.